Source: Research Gate
Conclusions Hydroxychloroquine has been shown to have consistent clinical efficacy for COVID-19 when it is used early in the outpatient setting, and in general would appear to work better the earlier it is used. Overall HCQ is effective against COVID-19.
There is no credible evidence that HCQ results in worsening of COVID-19. HCQ has been shown to be safe for the treatment of COVID-19 when responsibly used.
TY – BOOK
AU – Prodromos, Chadwick
AU – Rumschlag, Tobias
PY – 2020/09/04
SP –
T1 – Hydroxychloroquine is Effective and Safe for the Treatment of COVID-19, and May be Universally Effective When Used Early Before Hospitalization: A Systematic Review
DO – 10.13140/RG.2.2.29781.65765
ER –
Abstract and Figures
INTROUCTION Hydroxychloroquine (HCQ) has shown efficacy against COVID-19 in some but not all studies. We hypothesized that systematic review would show HCQ to be: effective against COVID-19, more effective used earlier, not associated with worsening, and safe.
METHODS We searched PubMed, Cochrane, EmBase, Google Scholar, and Google for all reports on hydroxychloroquine as a treatment for COVID-19 patients. This included pre-prints and preliminary reports on larger COVID-19 studies. We examined the studies for efficacy, time of administration and safety.
RESULTS HCQ was found consistently effective against COVID-19 when used early, in the outpatient setting. It was found overall effective. No credible study found worse outcomes with HCQ use. No mortality or other serious safety issue was found
CONCLUSIONS HCQ is consistently effective against COVID-19 when used early in the outpatient setting, it is overall effective against COVID-19, it has not produced worsening, it is safe.
Introduction
There is a need for effective treatment for COVID-19 infection. Hydroxychloroquine (HCQ), with or without azithromycin, has been found to have efficacy as a treatment for COVID-19 in some studies [1, 2], while other studies have not shown efficacy[3, 4].
Some physicians have stated that HCQ has greater efficacy if given earlier in the course of the disease[5, 6]. Several studies showing negative efficacy have been withdrawn due to methodological improprieties [7].
We hypothesized that HCQ clinical studies would show significant efficacy more often than not for COVID-19; and that efficacy would be greater if HCQ were used earlier in the course of the disease. We also hypothesized that some studies that failed to show efficacy would be biased against positive efficacy and that no unbiased studies would show worsening.
We also hypothesized that HCQ would be found to be safe. Methods We searched PubMed, Cochrane, EmBase, Google Scholar, and Google for all reports on hydroxychloroquine as a treatment for COVID-19 patients. This included pre-prints and preliminary reports on larger COVID-19 studies. We included papers with HCQ alone as well as in combination with Azithramycin and/or Zinc. We excluded papers that studied Chloroquine. While Chloroquine has shown efficacy it has a higher side effects profile than HCQ. For this reason, and because HCQ is inexpensive and widely available we believe that future treatment will and should focus on HCQ. It was thus our priority to examine HCQ as fully as possible. We excluded papers that only examined hydroxychloroquine as a means to decrease transmission of coronavirus since our focus was on demonstrated clinical efficacy.
Reports were analyzed for efficacy, type of study, time of intervention with HCQ during the COVID-19 disease course, and for adverse events. Our final search was performed August 3rd, 2020.
Results
A total of 43 reports were found that examined hydroxychloroquine treatment for COVID-19 patients. 25 found positive clinical efficacy from using hydroxychloroquine for COVID-19 patients; 15 showed no improvement with HCQ, and 3 showed worse clinical results in patients who received HCQ. 11 of the studies found in our review examined HCQ efficacy on patients in the outpatient or “day hospital” and all reported positive results [8].
However in two of the studies [9, 10] the positive results, while clinically important (decreased risk of hospitalization and improvement in symptom resolution), were not statistically significant. We found 32 reports of HCQ treatment in hospitalized patients with COVID-19. Of these 32 reports of hospitalized patients, 14 reported good results, 15 reported no improvement and 3 reported worse results. 14 studies reported the time during treatment at which HCQ was initiated.
In nine studies HCQ was administered within 48 hours of admission. In six [11-16] of these nine, improvement was noted. In three it was not [3, 17, 18]. In five studies HCQ was administered more than 48 hours after admission or in the ICU. In two [19, 20] of these five improvement was noted. In three it was not [21-23]. In 18 studies the time of administration was not specified. Seven of the 43 total studies [12, 17, 20, 24-27] were chartless retrospective studies that used only billing codes.
These studies all allowed initiation of HCQ treatment at times that differed with initiation of the control treatment: with HCQ presumably being chosen at the physician’s discretion in worsening patients more in need of treatment. All such studies were felt to exhibit selection bias against a positive result. Four additional studies [9, 10, 15, 16] had positive trends toward efficacy that did not reach statistical significance.
In 1 study [22] 8% of the treatment group was untreated but not excluded from the treatment group calculations. In addition the median level of treatment was only 67% of the specified treatment. This large undertreatment of the treatment group was also felt to bias against a positive result. 19 of the 43 papers were pre-prints or otherwise not peer reviewed, 24 of the papers were from peer reviewed journals.
Of the eleven outpatient papers, all of which showed positive results, 7 were peer reviewed, 4 were not. Of the 32 hospitalization papers 17 were peer reviewed and 15 were not. Overall 12 of 24 or 50% of the peer reviewed papers, and 11 of 19 or 58% of the non-peer reviewed papers showed positive efficacy.
Some studies used HCQ alone, some had the addition of azithromycin or zinc. No outcome difference was seen with the addition of azithromycin (table 4). There were no deaths reported as a result of HCQ, azithromycin or Zinc treatment. Increased QTc was seen but not Torsades de Pointes. Adverse events that were felt to be likely due to HCQ treatment were non-life threatening. All were generally self limited adverse events that typically occur with HCQ. No permanent sequelae were described. Adverse events are listed in Tables 1-3.
Table 1
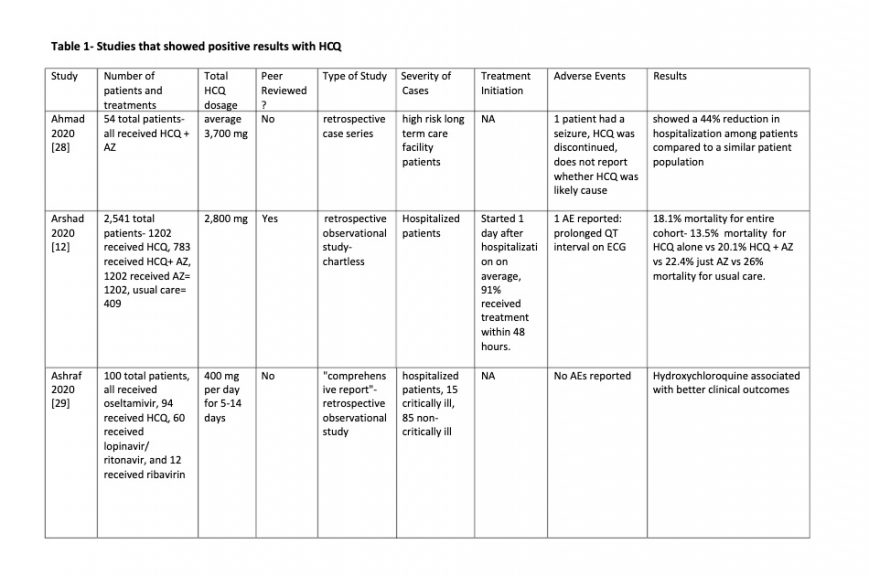





Table 2





Table 3

Table 4

Table 5

Discussion
This study has four important findings. The first is that HCQ appears to be consistently effective for the treatment of COVID-19 when used early in the course of disease in the outpatient setting, and is generally more effective the earlier it is used. The second is that overall HCQ has had efficacy against COVID-19 in a majority of studies. The third is that there are no unbiased studies showing a negative effect of HCQ treatment of COVID-19. The fourth is that HCQ appears to be safe for the treatment of COVID-19 when used responsibly.
TIMING OF HCQ USE: It was striking that 100% of the 11 of the studies which used HCQ early in the disease on an outpatient basis showed positive results. In two of the studies [9, 10] the benefit was only a trend. However the effects were clinically important: in Mitja’s study resolution of symptoms was decreased from 12 to 10 days; In Skipper’s study the rate of hospitalization was decreased by 60%. It is likely that with higher powering statistical significance would have been reached. In the 32 other studies HCQ was given on an inpatient basis with more advanced disease. The studies were divided into early, late and ICU administration times. The early use, within 48 hours of admission showed 6 of 9 or 67% of the studies to have positive efficacy. The two later groups, after 48 hours admission and in the ICU showed 2 of 5 or 40% to have positive efficacy. Thus, from 100% for early outpatient, to 67% for early hospital, to 40% for later hospital use, there appears to be a relationship with time of initiation of treatment, and better results the earlier HCQ is used.
OVERALL EFFICACY: 23 of the 43 studies (53%) showed a definite positive effect of HCQ vs COVID-19. However if negatively biased studies are removed and the clinically important positive trends from underpowered studies are moved to the positive efficacy group the ratio changes to 28 positive vs 9 no effect: a 75% positivity ratio of positive HCQ studies. Interestingly none of the no-effect studies showed a clear trend toward worsening.
RANDOMIZED CONTROLLED STUDIES (RCTs): Of the seven RCTs two [9, 10] were in the outpatient early treated group. As described above both studies had clinically important trends toward positive results, although were underpowered and did not reach statistical significance. The other five RCTs were in hospitalized patients later in disease where efficacy seems to be less. There was 1 positive [11], 3 no-effect [4, 43, 44], and 1 negative effect [22] studies. The negative effect study, however, was biased, as described below (“negative effect studies”), such that any negative or no-effect result would not be valid. Thus two of two RCTs with early treatment showed positive results, and one of three hospitalized patients had a positive result, consistent with the general finding of better results with earlier use.
NEGATIVE EFFECT STUDIES: Three studies had data that seemed to show worse outcomes with HCQ use. However had significant biases. And all were in hospitalized patients when results with HCQ are less good. Two [3, 16] of the three studies were well done studies that were nonetheless constrained by being chartless hospitalization studies that only used billing codes at particular time points to evaluate patients, but had no information as to events between these time points within their hospital course which led to initiation of treatment. Both were retrospective. Patients were not randomized to treatment with HCQ versus other care.
Rather patients apparently received HCQ at the discretion of the physician The time of administration of HCQ in the patients who received it was not specified during the hospitalization. This introduces selection bias in both studies toward treatment with HCQ for sicker patients who were faring worse after admission who presumably would be more likely to have treatment selected by their physician.
Attempting to normalize co-morbidities does not correct this bias because clinical progress of COVID-19 infection is not well predicted by pre-existing co-morbidities. This selection basis means patients who worsened after admission who are thereby more likely to have worse outcomes would be over represented in the HCQ treatment group.
For this reason negative results from the treatment arm of these studies are not valid because outcomes are moved negatively. A positive effect however would have validity since it could only occur despite the negative selection bias, not because of it.
The third study showing worse results with HCQ was a highly powered non-peer reviewed study whose primary outcome of 28 day mortality actually showed no difference between the HCQ treated group and the usual treatment group. Two of the secondary results did just barely reach significance negatively. [22]. However the reporting of results was flawed as follows. 8% of the treatment group patients did not receive HCQ at all; and the median number of days of treatment for all treated patients was only 6 out of a prescribed 9.
These facts mean that less than half of patients received the full treatment regimen or even two thirds of the full treatment regimen, with 1 in 12 receiving no treatment at all. These untreated and undertreated patient outcomes were however grouped with the fully treated patient outcomes.
If HCQ has any positive effect which we believe it is well established, this undertreatment would invalidate their borderline negative secondary results. In addition treatment was initiated more than 48 hours after admission when our aggregate data has shown a high incidence of no-effect results.
The study was not blinded introducing a potential undertreatment bias toward patients who were known by the staff to be treated with HCQ. This study most reasonably is actually a no effects study, which is common in already hospitalized patients such as these treated more than 48 hours after admission.
ADVERSE EVENTS: There have been fears among some that the increased QTc seen in some patients treated with HCQ or azithromycin would predispose to Torsades de Pointes (TDP) and then death from ventricular fibrillation. We found no such deaths, or death from any cause related to HCQ treatment, and indeed only 1 case of TDP at all – which resolved spontaneously without treatment and without sequelae.
All of the adverse events which seemed attributable to HCQ treatment in the 43 studies were typical side effects commonly seen with HCQ. These included nausea, vomiting, diarrhea, stomach pain, headache, rash, dizziness, itching and blurred vision. In all cases there was no indication of persistence of symptoms after discontinuance of the HCQ.
HCQ has been used with great safety for more than 50 years, and the relatively minor adverse events seen in these studies is consistent with this high safety profile.
STRENGTHS AND WEAKNESSES: A strength of this study is the large number of cohorts. A further strength is the critical methodological study analysis heretofore not attempted to our knowledge for these studies.
One weakness is the heterogeneity of study designs which rendered comparison of study results challenging. Another perceived weakness of the study could be that these include reports made outside of peer-reviewed literature. Multiple papers reporting both improvement and no efficacy using hydroxychloroquine that have been included in the study are either pre-prints or preliminary results of larger trials.
Because of the unprecedented and time sensitive nature of the SARS-COV2 pandemic the scientific community has shared data and studies on a level unseen prior to this emergency. We believe that these reports hold valuable information and decided to include them regardless of the way in which they were published.
In addition we found that both the peer-reviewed and non-peer reviewed papers showed a similar breakdown between studies showing efficacy vs not so that bias was not introduced.
SIGNIFICANCE: We believe our findings have substantial societal global importance since there have been numerous edicts either preventing HCQ use for COVID-19 or limiting it to the inpatient setting which we believe have resulted in many unnecessary deaths.
Our findings showing efficacy and safety of HCQ against COVID-19 indicate that HCQ should be freely available to patients and physicians who choose to use it. And it should especially be freely available to be used on an outpatient basis before hospitalization where it appears to be more effective and where early fears of fatal heart arrhythmias have been shown to be unfounded[45].
This is particularly important because the only drug to show efficacy, Remdesivir, has shown no significant benefit in a recent study [46].It is also expensive and not widely available. Convalescent plasma has shown benefit [47] but even this is not well validated and plasma is not available in large numbers of doses.
Thus HCQ with proven efficacy and safety, a cost of 37 cents per pill and thus a total treatment cost of under 20 dollars[48], versus 3,100 dollars for Remdesivir[49], as well as wide supply chain availability, would appear to be the best COVID-19 treatment option available and needs to be widely promoted as such.
Unfortunately the controversies surrounding HCQ have resulted in physicians being afraid to prescribe it for reasons which have nothing to do with medicine, and in patients being afraid to take it due to spurious reports of danger, or fears that is not effective.
It is hoped that this study will disabuse the medical community of these misapprehensions about efficacy and validate that it is both efficacious and safe, and needs to be freely prescribable. Thousands of lives may lie in the balance.
We also do not believe that randomized controlled studies are necessary before HCQ is authorized for general use because the efficacy seen in studies already done indicates that control patients in such studies might die unnecessarily; and because the time delay to do any such study would cause yet more deaths by preventing HCQ use when it is most needed – which is immediately.
Our study has shown that good evidence of efficacy exists; and there is no safety, cost, or supply reason to not treat now. Unnecessary death from delayed treatment is too high a price to pay for greater certainty of knowledge.
Many may have already died unnecessarily due to HCQ misinformation and it is imperative that we do not further add to the toll.
Conclusions Hydroxychloroquine has been shown to have consistent clinical efficacy for COVID-19 when it is used early in the outpatient setting, and in general would appear to work better the earlier it is used. Overall HCQ is effective against COVID-19.
There is no credible evidence that HCQ results in worsening of COVID-19. HCQ has been shown to be safe for the treatment of COVID-19 when responsibly used.
References
[1] A.d.A. Monforte, A. Tavelli, F. Bai, G. Marchetti, A. Cozzi-Lepri, Effectiveness of Hydroxychloroquine in COVID-19 disease: A done and dusted situation?, International Journal of Infectious Diseases, (1920).
[2] V. Guérin, P. Lévy, J.-L. Thomas, T. Lardenois, P. Lacrosse, E. Sarrazin, N.R. de Andreis, M. Wonner, Azithromycin and hydroxychloroquine accelerate recovery of outpatients with mild/moderate COVID-19, Asian Journal of Medicine and Health, 18 (2020) 45-55.
[3] J. Mallat, F. Hamed, M. Balkis, M.A. Mohamed, M. Mooty, A. Malik, A. Nusair, F. Bonilla, Hydroxychloroquine is associated with slower viral clearance in clinical COVID-19 patients with mild to moderate disease: A retrospective study, medRxiv, (2020).
[4] A.B. Cavalcanti, F.G. Zampieri, L.C. Azevedo, R.G. Rosa, A. Avezum, V.C. Veiga, R.D. Lopes, L. Kawano-Dourado, L.P. Damiani, A.J. Pereira, Hydroxychloroquine alone or in combination with azithromycin to prevent major clinical events in hospitalised patients with coronavirus infection (COVID-19): rationale and design of a randomised, controlled clinical trial, medRxiv, (2020).
[5] V. Zelenko, Nunesfarma, www.nunesfarma.com.br, 2020, pp. 2.
[6] M. Million, J. Lagier, P. Gautret, P. Colson, P. Fournier, S. Amrane, M. Hocquart, M. Mailhe, V. Esteves-Vieira, B. Doudier, Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: A retrospective analysis of 1061 cases in Marseille, France. Travel Med Infect Dis101738, 2020.
[7] J.F. Gumbrecht, Maggie, Two Coronavirus studies retracted after questions emerge about data, CNN.com, 2020.
[8] R.B. Esper, R.S. da Silva, F. Oikawa, M. Castro, A. Razuk-Filho, P. Junior, Empirical treatment with hydroxychloroquine and azithromycin for suspected cases of COVID-19 followed-up by telemedicine, Prevent Senior Institute SP, Brazil, ed. São Paulo, 25 (2020).
[9] O. Mitjà, M. Corbacho-Monné, M. Ubals, C. Tebe, J. Peñafiel, A. Tobias, E. Ballana, A. Alemany, N. Riera-Martí, C.A. Pérez, C. Suñer, P. Laporte, P. Admella, J. Mitjà, M. Clua, L. Bertran, M. Sarquella, S. Gavilán, J. Ara, J.M. Argimon, J. Casabona, G. Cuatrecasas, P. Cañadas, A. Elizalde-Torrent, R. Fabregat, M. Farré, A. Forcada, G. Flores-Mateo, E. Muntada, N. Nadal, S. Narejos, A.N. Gil-Ortega, N. Prat, J. Puig, C. Quiñones, J. Reyes-Ureña, F. Ramírez-Viaplana, L. Ruiz, E. Riveira-Muñoz, A. Sierra, C. Velasco, R.M. Vivanco-Hidalgo, A. Sentís, G.B. C, B. Clotet, M. Vall-Mayans, Hydroxychloroquine for Early Treatment of Adults with Mild Covid-19: A Randomized-Controlled Trial, Clin Infect Dis, (2020).
[10] C.P. Skipper, K.A. Pastick, N.W. Engen, A.S. Bangdiwala, M. Abassi, S.M. Lofgren, D.A. Williams, E.C. Okafor, M.F. Pullen, M.R. Nicol, Hydroxychloroquine in nonhospitalized adults with early COVID-19: a randomized trial, Annals of internal medicine, (2020).
[11] Z. Chen, J. Hu, Z. Zhang, S. Jiang, S. Han, D. Yan, R. Zhuang, B. Hu, Z. Zhang, Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial, MedRxiv, (2020).
[12] S. Arshad, P. Kilgore, Z.S. Chaudhry, G. Jacobsen, D.D. Wang, K. Huitsing, I. Brar, G.J. Alangaden, M.S. Ramesh, J.E. McKinnon, Treatment with hydroxychloroquine, azithromycin, and combination in patients hospitalized with COVID-19, International Journal of Infectious Diseases, (2020).
[13] B. Davido, G. Boussaid, I. Vaugier, T. Lansaman, F. Bouchand, C. Lawrence, J.-C. Alvarez, P. Moine, V. Perronne, F. Barbot, nImpact of medical care including anti-infective agents use on the prognosis of COVID-19 hospitalized patients over time, International Journal of Antimicrobial Agents, (2020) 106129.
[14] H. Xue, Y. Liu, P. Luo, X. Liu, L. Qiu, D. Liu, J. Li, Hydroxychloroquine treatment in COVID-19: a descriptive observational analysis of 30 cases from a single center in Wuhan, China, Journal of Medical Virology, (2020).
[15] O. Paccoud, F. Tubach, A. Baptiste, A. Bleibtreu, D. Hajage, G. Monsel, G. Tebano, D. Boutolleau, E. Klement, N. Godefroy, Compassionate use of hydroxychloroquine in clinical practice for patients with mild to severe Covid-19 in a French university hospital, Clinical Infectious Diseases, (2020).
[16] M. Mahevas, V.-T. Tran, M. Roumier, A. Chabrol, R. Paule, C. Guillaud, S. Gallien, R. Lepeule, T.-A. Szwebel, X. Lescure, No evidence of clinical efficacy of hydroxychloroquine in patients hospitalized for COVID-19 infection with oxygen requirement: results of a study using routinely collected data to emulate a target trial, MedRxiv, (2020).
[17] J. Geleris, Y. Sun, J. Platt, J. Zucker, M. Baldwin, G. Hripcsak, A. Labella, D.K. Manson, C. Kubin, R.G. Barr, Observational study of hydroxychloroquine in hospitalized patients with Covid-19, New England Journal of Medicine, (2020).
[18] E.S. Rosenberg, E.M. Dufort, T. Udo, L.A. Wilberschied, J. Kumar, J. Tesoriero, P. Weinberg, J. Kirkwood, A. Muse, J. DeHovitz, Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York state, Jama, (2020).
[19] B. Yu, C. Li, P. Chen, J. Li, H. Jiang, D.-W. Wang, Beneficial effects exerted by hydroxychloroquine in treating COVID-19 patients via protecting multiple organs, Science China Life Sciences, (2020) 1-4.
[20] B. Yu, C. Li, P. Chen, N. Zhou, L. Wang, J. Li, H. Jiang, D.-W. Wang, Low dose of hydroxychloroquine reduces fatality of critically ill patients with COVID-19, Science China Life Sciences, (2020) 1-7.
[21] M.H. An, M.S. Kim, B.-O. Kim, S.H. Kang, W.J. Kimn, S.K. Park, H.-W. Park, W. Yang, J. Jang, S. Jang, Treatment Response to Hydroxychloroquine and Antibiotics for mild to moderate COVID-19: a retrospective cohort study from South Korea, medRxiv, (2020).
[22] P. Horby, M. Mafham, L. Linsell, J.L. Bell, N. Staplin, J.R. Emberson, M. Wiselka, A. Ustianowski, E. Elmahi, B. Prudon, Effect of Hydroxychloroquine in Hospitalized Patients with COVID-19: Preliminary results from a multi-centre, randomized, controlled trial, medRxiv, (2020).
[23] A. Lopez, G. Duclos, B. Pastene, K. Bezulier, R. Guilhaumou, C. Solas, L. Zieleskiewicz, M. Leone, Effects of Hydroxychloroquine on Covid-19 in Intensive Care Unit Patients: Preliminary Results, International Journal of Antimicrobial Agents, (2020) 106136.
[24] A. Ip, D.A. Berry, E. Hansen, A.H. Goy, A.L. Pecora, B.A. Sinclaire, U. Bednarz, M. Marafelias, S.M. Berry, N.S. Berry, Hydroxychloroquine and Tocilizumab Therapy in COVID-19 Patients-An Observational Study, medRxiv, (2020).
[25] S. Singh, A. Khan, M. Chowdhry, A. Chatterjee, Outcomes of Hydroxychloroquine Treatment Among Hospitalized COVID-19 Patients in the United States-Real-World Evidence From a Federated Electronic Medical Record Network, medRxiv, (2020).
[26] J. Magagnoli, S. Narendran, F. Pereira, T.H. Cummings, J.W. Hardin, S.S. Sutton, J. Ambati, Outcomes of hydroxychloroquine usage in United States veterans hospitalized with Covid-19, Med, (2020).
[27] D.R. Rivera, S. Peters, O.A. Panagiotou, D.P. Shah, N.M. Kuderer, C.-Y. Hsu, S.M. Rubinstein, B.J. Lee, T.K. Choueiri, G. de Lima Lopes, Utilization of COVID-19 treatments and clinical outcomes among patients with cancer: A COVID-19 and Cancer Consortium (CCC19) cohort study, Cancer Discovery, (2020).
[28] I. Ahmad, M. Alam, R. Saadi, S. Mahmud, E. Saadi, Doxycycline and Hydroxychloroquine as Treatment for High-Risk COVID-19 Patients: Experience from Case Series of 54 Patients in Long-Term Care Facilities, medRxiv, (2020).
[29] M.A. Ashraf, N. Shokouhi, E. Shirali, F. Davari-tanha, O. Memar, A. Kamalipour, A. Azarnoush, A. Mabadi, A. Ossareh, M. Sanginabadi, COVID-19 in Iran, a comprehensive investigation from exposure to treatment outcomes, medRxiv, (2020).
[30] N. Bernaola, R. Mena, A. Bernaola, A. Lara, C. Carballo, P. Larranaga, C. Bielza, Observational Study of the Efficiency of Treatments in Patients Hospitalized with Covid-19 in Madrid, medRxiv, (2020).
[31] P. Carlucci, T. Ahuja, C.M. Petrilli, H. Rajagopalan, S. Jones, J. Rahimian, Hydroxychloroquine and azithromycin plus zinc vs hydroxychloroquine and azithromycin alone: outcomes in hospitalized COVID-19 patients, medRxiv, (2020).
[32] F.J.M. de Novales, G. Ramírez-Olivencia, M. Estébanez, B. de Dios, M.D. Herrero, T. Mata, A.M. Borobia, C. Gutiérrez, M. Simón, A. Ochoa, Early hydroxychloroquine is associated with an increase of survival in COVID-19 patients: an observational study, (2020).
[33] P. Gautret, J.-C. Lagier, P. Parola, L. Meddeb, M. Mailhe, B. Doudier, J. Courjon, V. Giordanengo, V.E. Vieira, H.T. Dupont, Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial, International journal of antimicrobial agents, (2020) 105949.
[34] P. Gautret, J.-C. Lagier, P. Parola, L. Meddeb, J. Sevestre, M. Mailhe, B. Doudier, C. Aubry, S. Amrane, P. Seng, Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: A pilot observational study, Travel medicine and infectious disease, (2020) 101663.
[35] J.-W. Kim, E.J. Kim, H.H. Kwon, C.Y. Jung, K.C. Kim, J.-Y. Choe, H.-L. Hong, Lopinavir-ritonavir versus hydroxychloroquine for viral clearance and clinical improvement in patients with mild to moderate coronavirus disease 2019, The Korean journal of internal medicine, (2020).
[36] M.S. Kim, S.-W. Jang, Y.-K. Park, B.-o. Kim, T.-H. Hwang, S.H. Kang, W.J. Kim, H.-W. Park, W. Yang, J. Jang, Treatment Response to Hydroxychloroquine, Lopinavir/Ritonavir, and Antibiotics for Moderate COVID 19: A First Report on the Pharmacological Outcomes from South Korea, medRxiv, (2020).
[37] J.-C. Lagier, M. Million, P. Gautret, P. Colson, S. Cortaredona, A. Giraud-Gatineau, S. Honoré, J.-Y. Gaubert, P.-E. Fournier, H. Tissot-Dupont, Outcomes of 3,737 COVID-19 patients treated with hydroxychloroquine/azithromycin and other regimens in Marseille, France: A retrospective analysis, Travel medicine and infectious disease, (2020) 101791.
[38] E. Sbidian, J. Josse, G. Lemaitre, I. Mayer, M. Bernaux, A. Gramfort, N. Lapidus, N. Paris, A. Neuraz, I. Lerner, Hydroxychloroquine with or without azithromycin and in-hospital mortality or discharge in patients hospitalized for COVID-19 infection: a cohort study of 4,642 in-patients in France, medRxiv, (2020).
[39] M. Scholz, R. Derwand, V. Zelenko, COVID-19 outpatients–early risk-stratified treatment with zinc plus low dose hydroxychloroquine and azithromycin: a retrospective case series study, (2020).
[40] A. Giacomelli, G. Pagani, A.L. Ridolfo, A. Oreni, F. Conti, L. Pezzati, L. Bradanini, G. Casalini, S. Passerini, A. Torre, Early administration of lopinavir/ritonavir plus hydroxychloroquine does not alter the clinical course of SARS-CoV-2 infection: a retrospective cohort study, medRxiv, (2020).
[41] M. Kalligeros, F. Shehadeh, E. Atalla, E.K. Mylona, S. Aung, A. Pandita, J. Larkin, M. Sanchez, F. Touzard-Romo, A. Brotherton, Hydroxychloroquine use in Hospitalized Patients with COVID-19: An observational matched cohort study, Journal of Global Antimicrobial Resistance, (2020).
[42] J.M. Molina, C. Delaugerre, J. Le Goff, B. Mela-Lima, D. Ponscarme, L. Goldwirt, N. de Castro, No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection, Med Mal Infect, 50 (2020) 30085-30088.
[43] NIH halts clinical trial of hydroxychloroquine, National Institutes of Health, NIH.gov, 2020.
[44] W. Tang, Z. Cao, M. Han, Z. Wang, J. Chen, W. Sun, Y. Wu, W. Xiao, S. Liu, E. Chen, W. Chen, X. Wang, J. Yang, J. Lin, Q. Zhao, Y. Yan, Z. Xie, D. Li, Y. Yang, L. Liu, J. Qu, G. Ning, G. Shi, Q. Xie, Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: open label, randomised controlled trial, Bmj, 369 (2020) m1849.
[45] C.C. Prodromos, Hydroxychloroquine is protective to the heart, not Harmful: A systematic review, New Microbes and New Infections, (2020) 100747.
[46] C.D. Spinner, R.L. Gottlieb, G.J. Criner, J.R.A. López, A.M. Cattelan, A.S. Viladomiu, O. Ogbuagu, P. Malhotra, K.M. Mullane, A. Castagna, Effect of Remdesivir vs Standard Care on Clinical Status at 11 Days in Patients With Moderate COVID-19: A Randomized Clinical Trial, JAMA, (2020).
[47] K. Duan, B. Liu, C. Li, H. Zhang, T. Yu, J. Qu, M. Zhou, L. Chen, S. Meng, Y. Hu, Effectiveness of convalescent plasma therapy in severe COVID-19 patients, Proceedings of the National Academy of Sciences, 117 (2020) 9490-9496.
[48] Price Guide- Hydroxychloroquine Prices, Coupons, and Patient Assitance Programs, Drugs.com, 2020.
[49] S. Lupkin, Remdesivir Priced At More Than $3,100 For A Course Of Treatment, National Public Radio, npr.com, 2020.

