Source: HCQ Meta
HCQ is effective for COVID-19. The probability that an ineffective treatment generated results as positive as the 115 studies to date is estimated to be 1 in 20 million (p = 0.000000049).
•Early treatment is most successful, with 100% of studies reporting a positive effect and an estimated reduction of 63% in the effect measured (death, hospitalization, etc.) using a random effects meta-analysis, RR 0.37 [0.30-0.47].
•100% of Randomized Controlled Trials (RCTs) for early, PrEP, or PEP treatment report positive effects, the probability of this happening for an ineffective treatment is 0.002.
•There is evidence of bias towards publishing negative results. Significantly more retrospective studies report negative results compared to prospective studies, p = 0.05.
•Significantly more studies in North America report negative results compared to the rest of the world, p = 0.005.
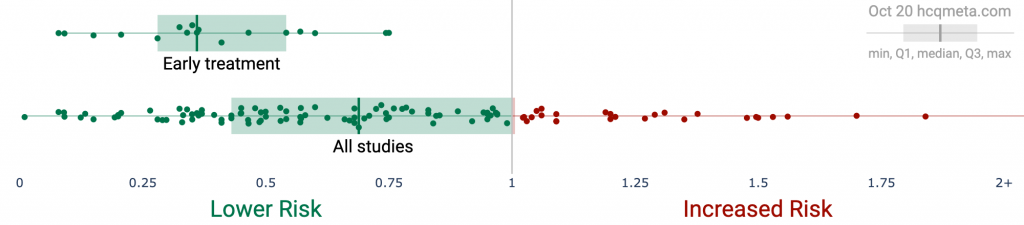
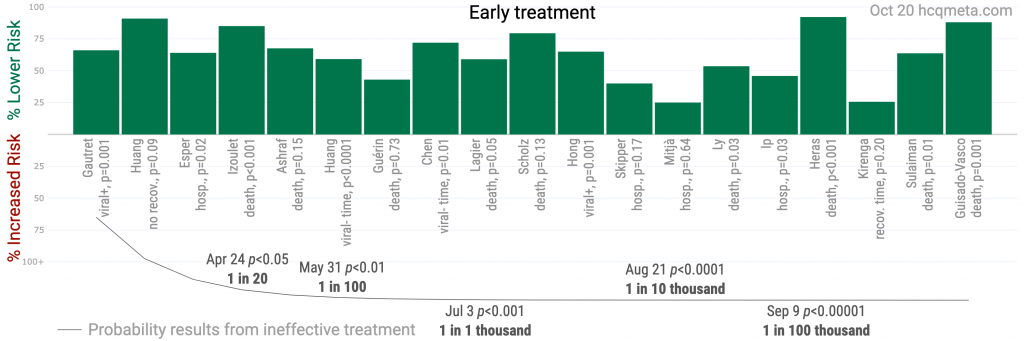

Figure 1. A. Scatter plot showing the distribution of effects reported in early treatment studies and in all studies. Early treatment is more effective. B and C. Study results ordered by date, with the line showing the probability that the observed frequency of positive results occurred due to random chance from an ineffective treatment.
Introduction
We analyze all significant studies concerning the use of HCQ (or CQ) for COVID-19 (Appendix 1), showing the effect size and associated p value for results comparing to a control group. Typical meta analyses involve subjective selection criteria and bias evaluation, requiring an understanding of the criteria and the accuracy of the evaluations. However, the volume of studies presents an opportunity for a simple and transparent analysis aimed at detecting efficacy.If treatment was not effective, the observed effects would be randomly distributed (or more likely to be negative if treatment is harmful). We can compute the probability that the observed percentage of positive results (or higher) could occur due to chance with an ineffective treatment (the probability of >= k heads in n coin tosses, or the one-sided sign test / binomial test). Analysis of publication bias is important and adjustments may be needed if there is a bias toward publishing positive results. For HCQ, we find evidence of a bias toward publishing negative results.

Figure 2 shows stages of possible treatment for COVID-19. Pre-Exposure Prophylaxis (PrEP) refers to regularly taking medication before being infected, in order to prevent or minimize infection. In Post-Exposure Prophylaxis (PEP), medication is taken after exposure but before symptoms appear. Early Treatment refers to treatment immediately or soon after symptoms appear, while Late Treatment refers to more delayed treatment.ResultsFigure 3, Figure 4 and Table 1 show results by treatment stage, and Figure 5 shows a forest plot for a random effects meta-analysis of all studies. Analysis excluding studies with major issues is in Appendix 2.Early treatment. 100% of early treatment studies report a positive effect, with an estimated reduction of 63% in the effect measured (death, hospitalization, etc.) from the random effects meta-analysis, RR 0.37 [0.30-0.47].Late treatment. Late treatment studies are mixed, with 68% showing positive effects, and an estimated reduction of 22% in the random effects meta-analysis. Negative studies mostly fall into the following categories: they show evidence of significant unadjusted confounding, including confounding by indication; usage is extremely late; or they use an excessively high dosage.Pre-Exposure Prophylaxis. 74% of PrEP studies are positive, with an estimated reduction of 41% in the random effects meta-analysis. Negative studies are all studies of systemic autoimmune disease patients which either do not adjust for the different baseline risk of these patients at all, or do not adjust for the highly variable risk within these patients.Post-Exposure Prophylaxis. 100% of PEP studies are positive, with an estimated reduction of 31% in the random effects meta-analysis.
| Treatment time | Number of positive studies | Total number of studies | Percentage of positive studies | Probability of an equal or greater percentage of positive results due to random chance | Random effects meta-analysis results |
| Early treatment | 19 | 19 | 100% | 0.0000019 1 in 524 thousand | 63% improvement RR 0.37 [0.30‑0.47] |
| Late treatment | 49 | 72 | 68.1% | 0.0015 1 in 680 | 22% improvement RR 0.78 [0.70‑0.87] |
| Pre‑Exposure Prophylaxis | 17 | 23 | 73.9% | 0.017 1 in 58 | 41% improvement RR 0.59 [0.43‑0.81] |
| Post‑Exposure Prophylaxis | 3 | 3 | 100% | 0.13 1 in 8 | 31% improvement RR 0.69 [0.46‑1.03] |
| All studies | 86 | 115 | 74.8% | 0.000000049 1 in 20 million | 32% improvement RR 0.68 [0.62‑0.75] |
Table 1. Results by treatment stage. 2 studies report results for a subset with early treatment, these are not included in the overall results.
